Foto: Harald Unterweger/Uniklinikum Erlangen
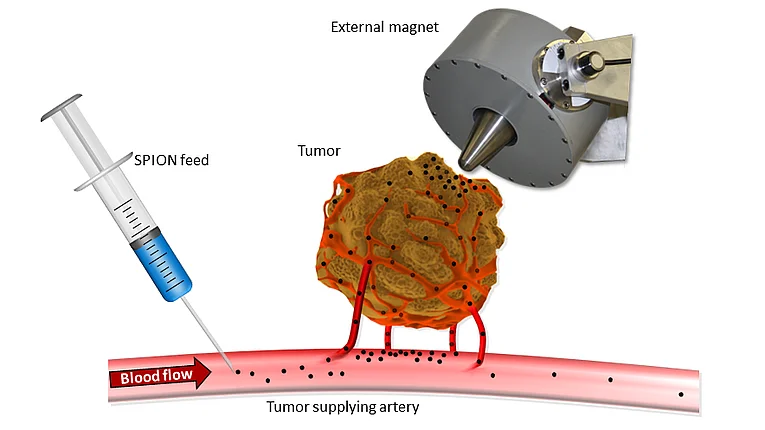
The starting point of all SEON activities is in the field of oncology. For good reason: cancer is one of the main causes of death worldwide and a disease that is associated with immense suffering for patients, but also for their social environment. Thus, there is a great need for new and innovative therapeutic options. Magnetic nanoparticles based on iron oxide have already generated great interest because of their potential medical applications as innovative therapeutic and diagnostic tools. In the magnetic enrichment of the nanoparticles in the tumor area, the so-called magnetic drug targeting (MDT), SEON takes the route of tumor-proximal, arterial application of the SPIONs into the tumor-supplying blood vessels, while the switched-on electromagnet is directly directed to the tumor area (see figure). This approach has the advantage over intravenous administration of greatly increased enhancement efficiency, which we have demonstrated in vitro and in vivo. We published the world's largest animal study on MDT to date back in 2013. In it, we showed that up to 35 times the dose of chemotherapeutic agent was found in the tumor area after a single MDT application compared to systemic administration. Furthermore, the study showed that in about 30% of the cases a complete tumor remission with few side effects was achieved.
Another advantage of this MDT concept is its connectivity to other forms of treatment and the associated exploitation of certain synergy effects. For example, it is possible to heat the magnetic particles using an alternating magnetic field and thereby perform local hyperthermia. We are also exploring the possibility of linking novel immunological therapies with MDT, for example by magnetically modifying T lymphocytes for tumor therapy.
Innovative diagnostics and therapy of liver tumors/metastases
Manfred-Roth Foundation, 2022-2025
Project goal
In order to demonstrate the performance of our particle formulation, which in many respects has already been carefully through-developed with iron oxide nanoparticles in the core and a cross-linked dextran shell, we plan to conduct animal experiments in a rat tumor model. This complex animal model will be established at SEON as part of this application, followed by imaging experiments with our radiology partners.
Cooperation partner
- Prof. Dr. Tobias Bäuerle
Development of a nanoparticle based anti-tumor gene therapy for treatment of breast cancer
DFG Funding, 2020-2023
Project goal
Our main goal in this research project is to develop a novel binary integrase (Int) system to specifically kill breast cancer cells. This system is based on a site-specific recombination reaction that activates specific expression in cancer cells only when it consists of two plasmids. One plasmid triggers expression of Int under the cancer-specific hTERT promoter. The second plasmid encodes a silenced gene substrate that can be activated by Int-catalyzed excision of a transcription terminator. For site-specific delivery, the plasmids are loaded onto SPIONs that can be directed to the desired site by magnetic forces. This is expected to greatly increase the intratumoral dose of the plasmids.
Cooperation partner
- Dr. Mikhail Kolot, Tel-Aviv University, Israel
Magnetic field applicator for local tumor therapy with nanoparticles (MAGTUNA)
ZIM-BMWi Funding, 2019-2020
Project goal
In this project, a novel therapy system is to be developed to minimize the side effects occurring during cancer therapy. It is a novel magnetic field applicator that will be used for the targeted control of nanoparticles. These particles are loaded with a chemotherapeutic agent and then brought specifically into the vicinity of the tumor by means of a magnetic field. With the help of this principle, it is possible to achieve a high local concentration of the active ingredient while sparing the entire organism. Another mode of operation of the applicator is to heat the particles near the tumor in order to trigger targeted destruction of the tumor tissue. Thanks to the combination of these two principles, it will be possible to achieve an effect that is locally limited mainly to the diseased tissue. At the same time, the surrounding healthy tissue is protected as far as possible.
Cooperation partner
- Senetics healthcare group GmbH & Co. KG, Erlangen
Integrative 'Big Data Modeling' for the development of novel therapeutic approaches for breast cancer (BIG-THERA)
Emerging Field Initiative, FAU, 2017-2019
Project goal
The main goal of BIG-THERA is to deepen our understanding of the interplay between the immune response and breast cancer by building an analytical platform that can significantly improve diagnosis and prognosis in the treatment of the disease. Within the grant period, EFI's BIG-THERA initiative will be able to provide proof-of-principle for each of the work packages outlined, namely, certifying cancer using imaging and OMICs techniques, establishing preclinical models in which immunological responses to human breast tumor cells can be measured at the molecular and imaging levels, and testing nanotechnology-based methods to increase lymphocyte content in tumors. In addition, BIG-THERA will be able to achieve initial results in synergistically linking imaging features and OMIC data to predict immune system involvement during cancer treatment. The implementation of Big Data in healthcare will be done with ethical standards in mind.
Cooperation partner
- Prof. Dr. Diana Dudziak, UK Erlangen
- Prof. Dr. Tobias Bäuerle, UK Erlangen
- Prof. Dr. Matthias Beckmann, UK Erlangen
- Prof. Dr. Peter Dabrock, FAU
- Prof. Dr. Peter Fasching, FAU
- Prof. Dr. Andreas Maier, FAU
- Prof. Dr. Falk Nimmerjahn, FAU
- Prof. Dr. Ana-Sunčana Smith, FAU
- Prof. Dr. Michael Uder, UK Erlangen
- Prof. Dr. Arnd Dörfler, UK Erlangen
- Prof. Dr. Tobias Engelhorn, UK Erlangen
- Prof. Dr. Abbas Agaimy, UK Erlangen
- Prof. Dr. Andriy Mokhir, FAU
- Prof. Dr. Helmut Ermert, Ruhr-Uni Bochum
- Prof. Dr. Martin F. Fromm, UK Erlangen
- Prof. Dr. Stefan Odenbach, TU Dresden
- Prof. Dr. Lutz Trahms, PTB Berlin
- Prof. Dr. Anette M. Schmidt, Universität zu Köln
- Prof. Dr. Urs Hafeli, University of British Columbia, Vancouver, Canada